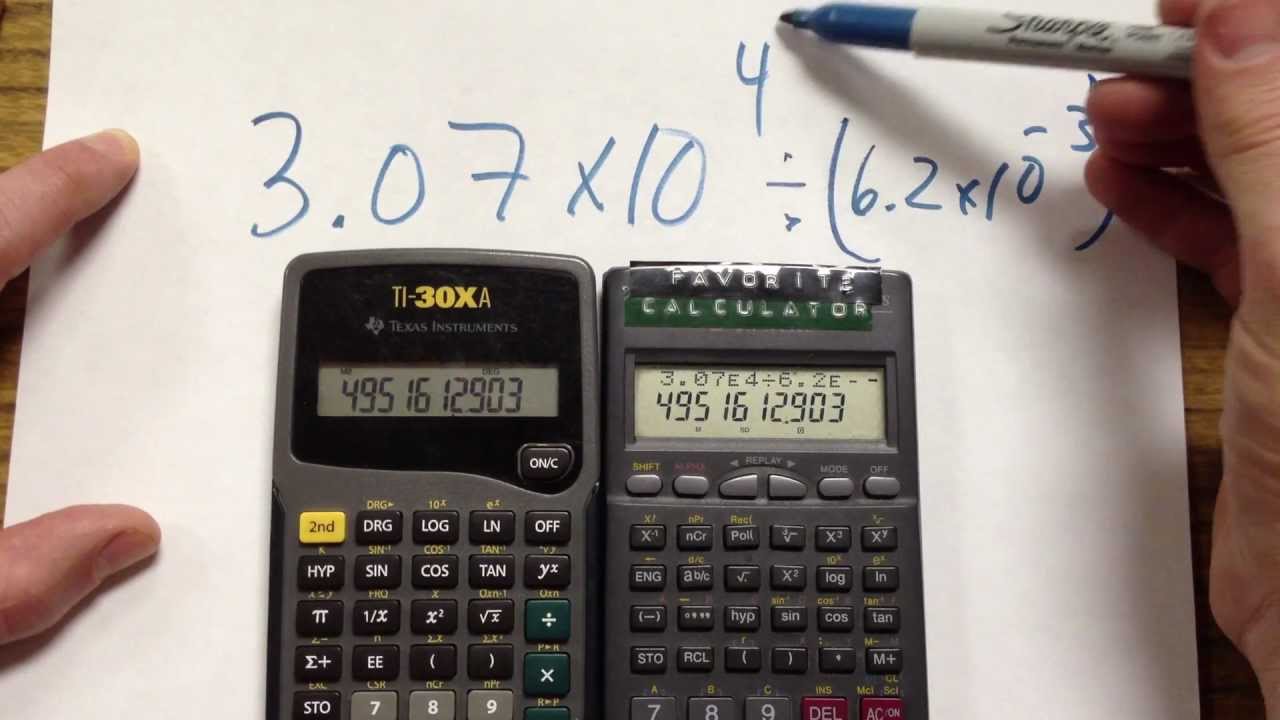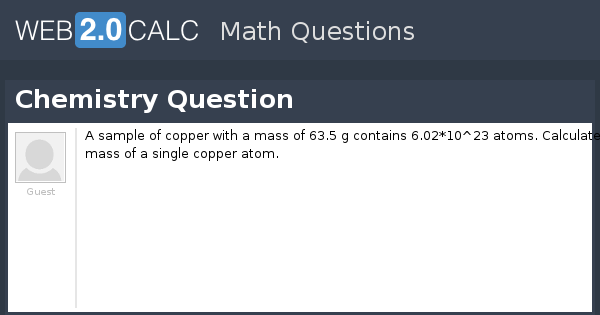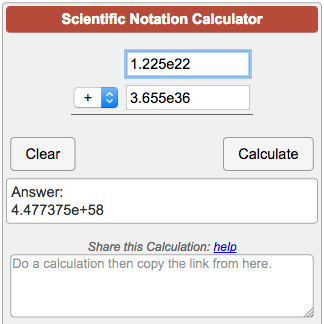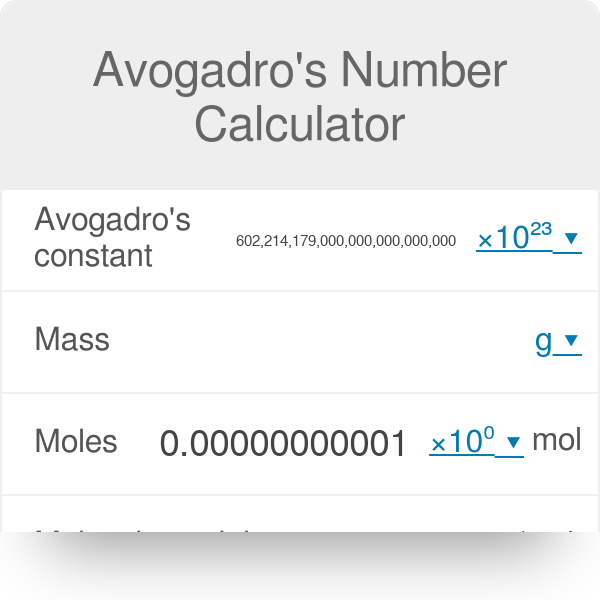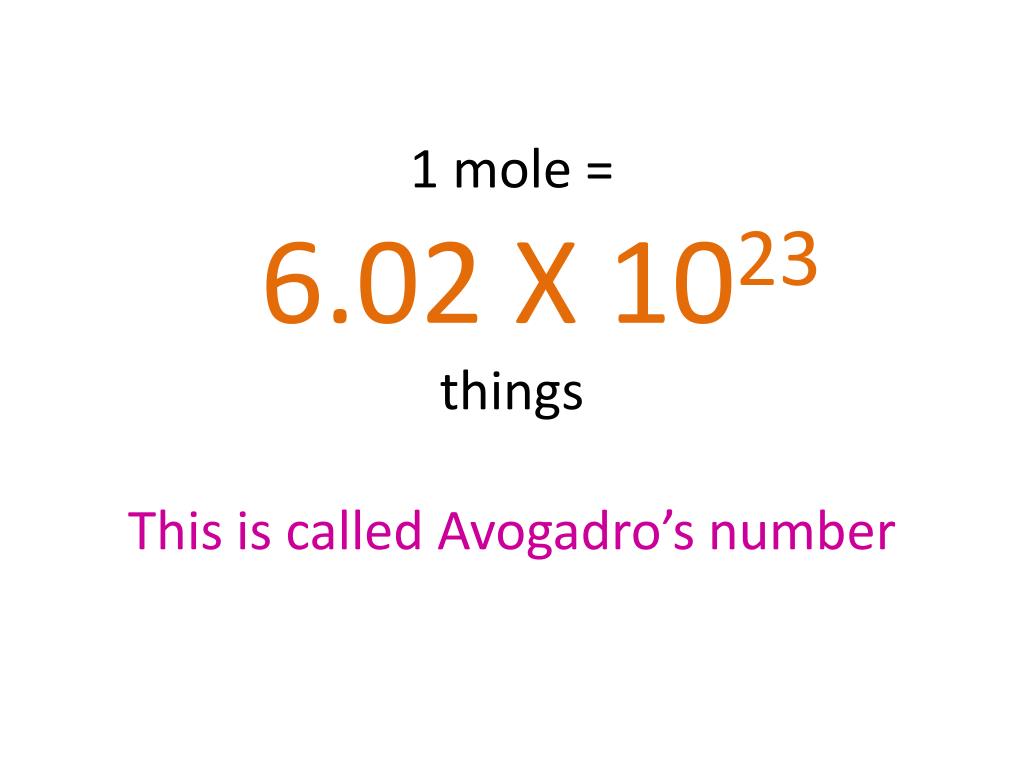
PPT - 1 mole = 6.02 X 10 23 things This is called Avogadro's number PowerPoint Presentation - ID:4272623

I need help with my calculator why is it showing this answer i dont know what i did wrong tho Im using a casio fx-991cw : r/calculators
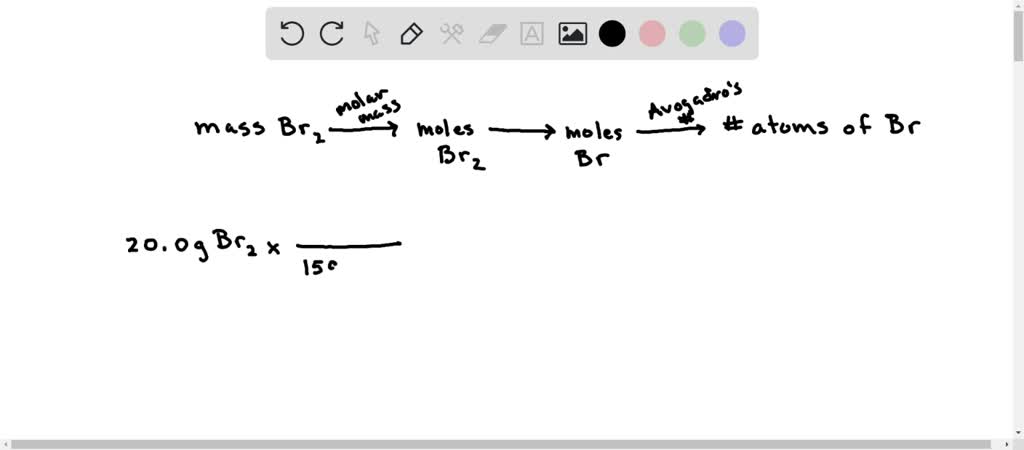
SOLVED: Calculate the number of Br atoms in 20.0g of liquid bromine (Br2). NOTE: Avogadro's number is 6.02 x 1023 A 7.53*10^23 B. 1.51*10^23 C. 3.01*10 ^23 D. 3.77*10^23 E. Nor correct response